| Journal of Clinical Gynecology and Obstetrics, ISSN 1927-1271 print, 1927-128X online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Clin Gynecol Obstet and Elmer Press Inc |
| Journal website http://www.jcgo.org |
Review
Volume 4, Number 2, June 2015, pages 197-202
Native Tissue Repair and Principles of Wound Healing: Introducing the Concept of Regenerative Surgery in Vaginal Prolapse Repair
Andri Nieuwoudta, Catherine Elizabeth Applebyb, c, Sambit Mukhopadhyayb
aDepartment of Obstetrics and Gynaecology, ZorgSaam Zeeuws-Vlaanderen, Wielingenlaan 2, 4535 PA Terneuzen, The Netherlands
bDepartment of Obstetrics and Gynaecology, Norfolk and Norwich University Hospital Trust, Colney Lane, Norwich NR4 7UY, UK
cCorresponding Author: Catherine Elizabeth Appleby, Department of Obstetrics and Gynaecology, Norfolk and Norwich University Hospital Trust, Colney Lane, Norwich NR4 7UY, UK
Manuscript accepted for publication June 12, 2015
Short title: Regenerative Surgery in Prolapse Repair
doi: http://dx.doi.org/10.14740/jcgo336w
- Abstract
- Introduction
- Wound Healing
- Regenerative Surgery
- Post-Operative Care and Wound Healing and Strength
- Native Tissue and Defect Specific Vaginal Prolapse Surgery
- Discussion
- Conclusion
- References
| Abstract | ▴Top |
Following the widespread use of vaginal synthetic mesh for vaginal prolapse repair and subsequent complications, native tissue vaginal surgery is once again gaining popularity. The traditional colporrhaphy utilizes native tissues only and has high reported failure rates. In this, the post vaginal synthetic mesh, era where native tissue alone is going to provide the primary building blocks, knowledge of how these tissues will react during wound healing is essential. This article reviews the principles of wound healing and regeneration of tissues in relation to native tissue vaginal reconstructive surgery.
Keywords: Regeneration; Wound healing; Regenerative surgery; Vaginal prolapse; Grafts; Regenerative medicine
| Introduction | ▴Top |
“When most types of human tissue are damaged, they repair themselves by forming a scar - a mechanically strong “patch” that restores structural integrity to the tissue without restoring physiological function. Much better, for a patient, would be like-for-like replacement of damaged tissue with something functionally equivalent”. Jamie Davies [1].
The art of medicine lies in the ability of the physician to make a diagnosis, and to treat the underlying abnormality, with a view to establishing morphological and functional normality. The basis of site-specific vaginal prolapse surgery includes identifying the defect and restoring the anatomical continuity using native tissue as building blocks.
The concept of restoring normal tissue function in humans started in the 1950s with organ transplant and then cell and tissue culture in the 1960s paved the way to tissue engineering [2]. From there stem cell therapy, use of regenerative factors, specific acellular scaffolds and therapeutic cloning have developed in order to restore the normal function of injured human tissues. William Heseltine first used “regenerative medicine” as a term to embrace all these fields [2], but the term has not been well defined [3]. An accepted definition is that regenerative medicine “replaces or regenerates human cells, tissue or organs, to restore or establish normal function” [3].
| Wound Healing | ▴Top |
Wound healing is a process that follows injury to tissues. The resulting level of function after healing depends on the tissue’s tendency to either scar or regenerate [4].
The following definite phases can be identified [5, 6]: 1) homeostatic phase; 2) inflammatory phase; 3) proliferation phase/scarring; 4) remodeling.
After initial homeostasis, a continual interaction occurs between “repair” and “remodeling” (Fig. 1). In the proliferation phase, the gap left by the injury is closed (repaired) as soon as possible with scar tissue, thus providing the tissue with some strength. On the other hand, with remodeling, there is a tendency to revert tissue back to the pre-injury model (anatomical and functional). These opposing actions are to a large extent controlled by the intensity and duration of the inflammatory phase. The inflammatory phase, in essence, is a clearing phase with removal of damaged tissue, and is driven by immunological reactions. The more intense this phase, the greater the tendency for scar tissue formation; the less intense, the greater the likelihood would be of remodeling with minimal scar formation.
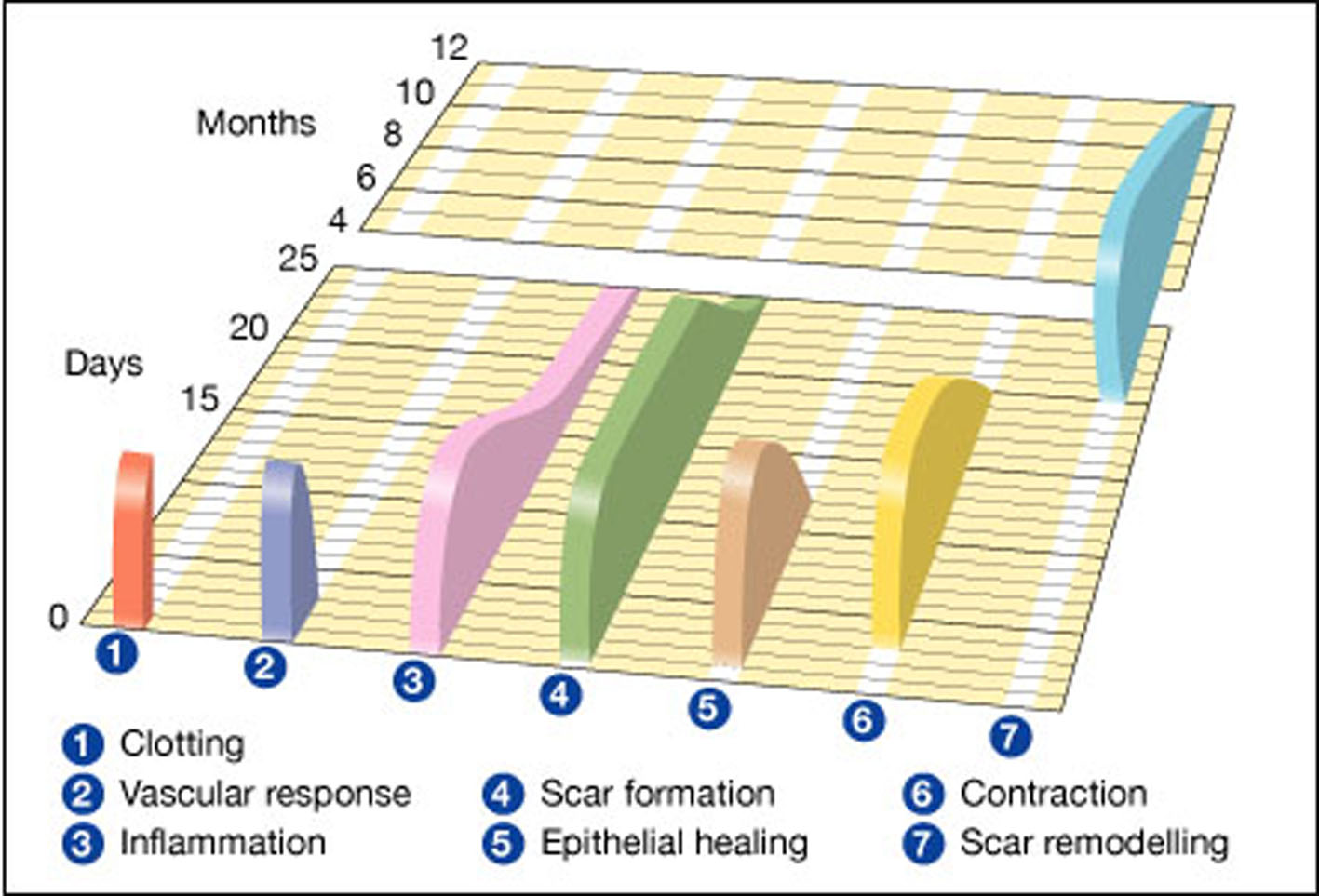 Click for large image | Figure 1. Wound healing phases against a timeline. 1 and 2 represent the hemostatic phase and 4, 5 and 6 represent the proliferation phase. Gregory S. Schultz, Glenn Ladwig and Annette Wysocki - adapted from Asmussen PD, Sollner B [6]. |
The time frame occupied by each phase will depend on the interaction between the inflammatory phase and the remodeling phase. Factors that drive the inflammatory phase (foreign materials, infections, tissue stress and ischemia, for example) will influence this.
Important molecular requirements must be present in order for the remodeling phase to result in regeneration to morphologically and functionally normal tissues. The driving forces behind this are the stem cells, either local or therapeutic. These multipotent cells are protected by the extracellular matrix (ECM), in which they are embedded [6, 7]. The ECM regulates cell behavior. The scaffold of the ECM is continually being broken down and restructured under the influence of a variety of enzymes: this activity is especially marked during wound healing [8]. Degradation of this protective shield is especially dependent on proteolytic cascades, which play a central role in the inflammatory phase of wound healing.
Homeostasis of the ECM structure is dependent on the presence of heparan sulphates - proteoglycans with a slightly negative charge [8]. By binding positively charged proteins such as growth factors, cytokines, cell surface proteins and ECM proteins, heparan sulphates isolate these proteins from degradation and stabilize the ECM scaffold. In the presence of inflammation heparan sulphates are degraded by proteolytic enzymes, resulting in disruption of the ECM and impaired healing.
It is with this knowledge that tissue engineering strives to replace damaged tissues with cells seeded on synthetic biodegradable structures, which mimic ECM in order to restore normal cellular function [9].
Heparan sulphate analogues (ReGeneraTing Agents - RGTA) are one of the early examples of regenerative medicine that reached daily clinical use [8]. The main difference between heparan sulphate and its analogue is that the analogue is resistant to proteolytic enzyme breakdown. When heparan sulphate is degraded at a wound site, the analogues can then bind to the available heparan sulphate binding proteins of the ECM. This enables normal cellular healing and tissue regeneration.
| Regenerative Surgery | ▴Top |
“Repair is an adaptation to loss of normal organ mass and leads to restoration of the continuity by synthesis of scar tissue without restoration to normal tissue. By contrast, regeneration restores the interrupted continuity by synthesis of the missing organ mass at the original anatomical site, yielding a regenerate. Regeneration restores the normal structure and function of an organ; repair does not”. Yannas [10].
In nature the range of healing is from total regeneration in some amphibians, to scar-free healing in early mammalian embryos to scarring in older mammals [11]. Through regenerative medicine one can take advantage of the potential of human cells to regenerate [12].
The ability of an organism to heal falls along a spectrum from regenerative, scar-free healing to scar-forming healing [2, 4, 11, 13]. The microenvironment of a wound influences its healing. Mammalian embryos heal without scarring. Compared with adult healing, there is a less intense, shorter inflammatory reaction to injury [4, 14]. The ECM structure of the restored dermis of an embryonic wound is the same as in normal skin, rather than in an adult wound where it is disordered. Scarring can be seen as a failure of the regeneration of the native tissue structure rather than a biochemical problem leading to formation of scar tissue [13]. In adult healing the more intense the inflammatory phase, the more intense the proliferation phase and the tendency to remodel with scar tissue formation. The less intense the inflammatory phase, the less remodeling will be towards scarring and more towards regeneration of normal tissue.
Young adults produced the maximum inflammatory response and scar the most, versus in older adults in whom the inflammatory response is reduced [11]. It is, however, the young who also regenerate better.
The difference between reparative and regenerative surgery relates to the reaction of native tissue to the surgical insult.
Native tissue regenerative surgery is the utilization of resident native tissue plus the manipulation of wound healing to enhance remodeling of damaged tissue to be morphologically and functionally normal. This is achieved by limiting the inflammatory phase of wound healing combined with support of new native tissue formation, for example by the utilization of biodegradable scaffolds [4]. Pharmacological support, good and safe surgical practices are also essential part of this process. In dentistry this is recognized as guided tissue regeneration (GTR) [15].
The key principle of regenerative surgery is to minimize tissue inflammation (Fig. 2). The technique used should induce less tissue trauma by following embryological dissection planes and align tissue layers in a tension free manner with splinting to keep it tension free during the healing process. The use of monofilament suture materials and avoidance of inflammatory inducing synthetic materials are essential. Biodegradable scaffolds give temporary support to the repair site limiting tissue tension and encouraging ingrowth of resident tissue stem cells [4].
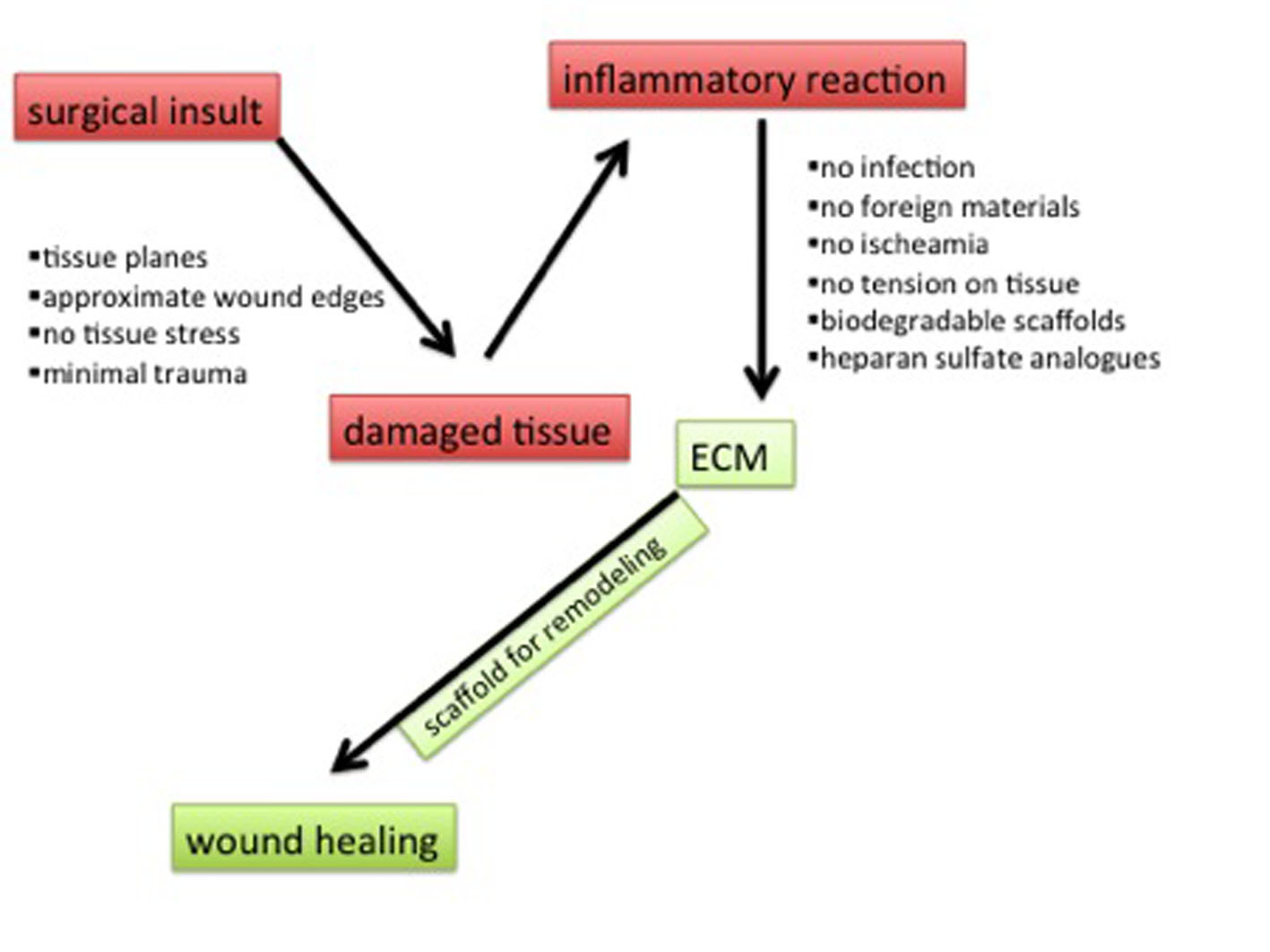 Click for large image | Figure 2. Surgical principles and influence on wound healing. The inflammatory reaction to a surgical insult can be limited by adherence to surgical principles to protect the extracellular matrix and therefore influence wound healing. |
| Post-Operative Care and Wound Healing and Strength | ▴Top |
The study of vaginal surgical wound strength and how it is influenced by the physical activities of the patient is central to post-operative care. Surgical outcome assessments can be timed with this knowledge. The post-operative care is traditionally focused on resting for the first 6 weeks post-operatively from basically all physical activity, with then increasing the strain that is put on the surgical field by increasing the activity of the patient.
The timeline of wound healing (Fig. 3) [16] illustrates that by 6 weeks the maturation and remodeling phase has only just started, with basically no increase of wound strength. It is only after 6 months that wound strength reaches 50% of normal tissue. By 1 year is there a completion of tissue maturation and remodeling, thus this would be the time to accurately predict the durability of the repair.
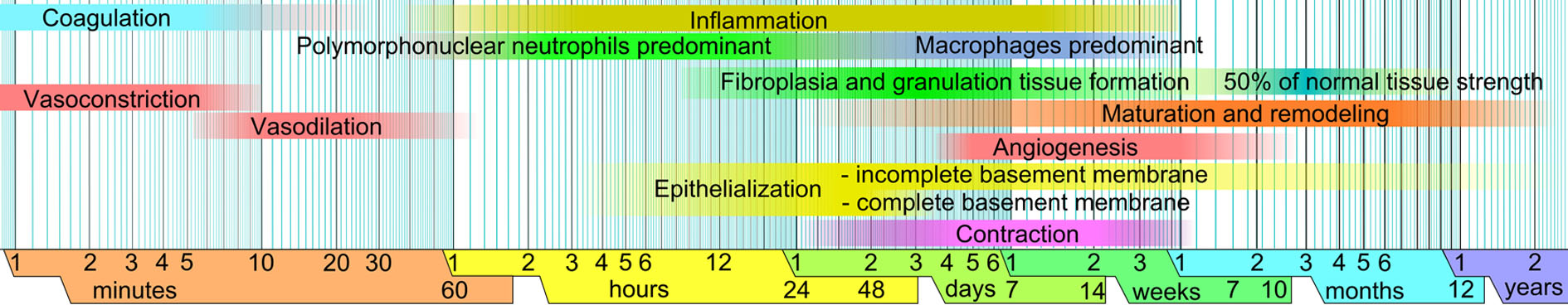 Click for large image | Figure 3. Timeline of wound healing. Six months after injury native tissue healing has only achieved 50% of normal tissue strength. Haggstrom [16]. |
Post-operative assessment should thus be: 1) 6 weeks: assessment of inflammatory phase intensity; 2) 6 months: assessment of inflammatory phase intensity and onset of remodeling/scarring. At this stage one can start to predict durability of wound strength; 3) 12 months: prediction of surgical outcome.
| Native Tissue and Defect Specific Vaginal Prolapse Surgery | ▴Top |
Defect-specific vaginal prolapse surgery includes defining the defect, or defects, that lead to the collapse of the vaginal support structures and strengthening these weakened areas of support.
The folding of fascial tissues during conventional colporrhaphies with resultant tension on the native tissues and scarring during wound healing cannot fall under the definition of regenerative surgery. Poor surgical outcome can be due to residual disease, or to recurrence of disease: both instances can explain suboptimal surgical outcomes of traditional anterior colporrhaphies. Midline plication means the native tissue is subjected to ischemia and tension during the surgical process, resulting in excessive scarring. This microenvironment counters the principles of regenerative medicine and does not allow regenerative healing. The underlying cause of the prolapsed vaginal wall is not addressed, merely treating the bulging wall: it is to an extent destructive surgery more than reconstructive surgery. This may explain why, even when traditional repair is enhanced with supportive regenerative principles, the recurrence rate remains high.
In defect-specific vaginal prolapse surgery, the primary surgery is aimed at dissecting the fascia free from the overlying vaginal mucosa, following embryological tissue planes, with minimal tissue damage during the surgical insult. This will enable the surgeon to accurately define the damaged areas and reconstruct the lateral vaginal supports and pericervical ring. At the defective areas there is a combination of a mechanical defect and biochemical defects of the surrounding fascial tissue [17]. When the repair of these areas is necessary the surgeon will have to deal with poor native tissue qualities in older age group; however, younger patients tend to have better quality of tissue. Another factor to be taken account includes suture lines being intermittently subjected to pressure changes from the abdominal cavity. The combination of dissection and repair is important in protecting the native tissue; dissection should avoid damaging the fascial sheets and repair should approximate the tissue without allowing tension on these fascial sheets. In the pelvis a third force is at play, variable tensions on the suture lines can lead to early breakdown of the repair or put tension on the native tissue and thereby stimulating the inflammatory reaction. To counter this, temporary splinting or support is required to keep the native tissue in place until proper strong collagen is being produced to strengthen the previous defective areas [18]. In regenerative surgery new tissue can be remodeled under the guidance of a biodegradable synthetic scaffold or biodegradable xenografts.
This is the meeting point between defect-specific surgery and regenerative wound healing practices: a true starting point for reconstructive surgery.
By treating the cause of the prolapsed vaginal wall, and not the symptom of the defective support system, i.e. the vaginal bulge, one can expect to improve surgical outcome. By augmenting and splinting the suture lines with biodegradable grafts, the tension is taken away from the native tissue (Fig. 4), with resultant improved tissue regeneration and healing without scarring.
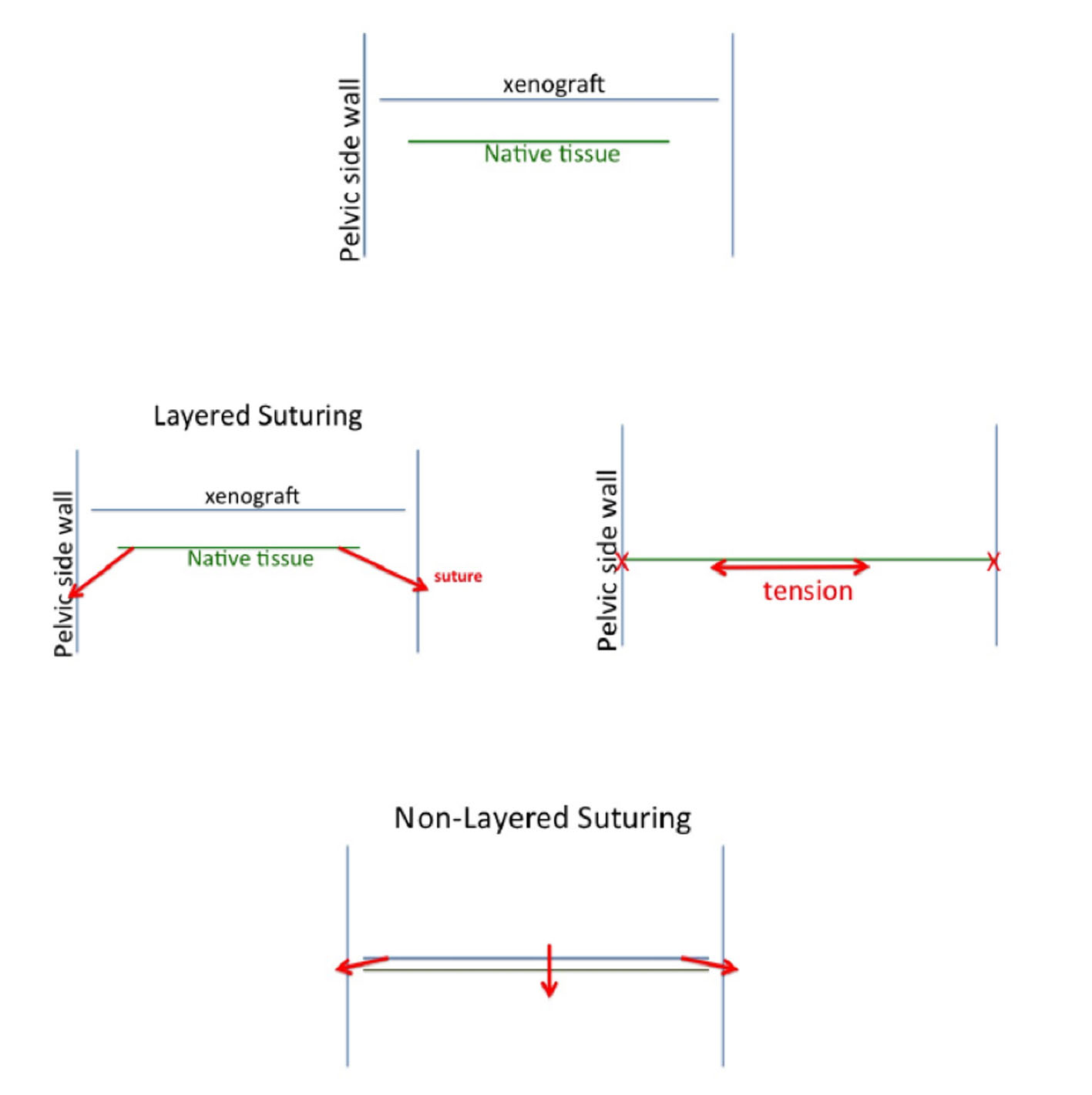 Click for large image | Figure 4. Tension free suturing. An example of how tissue tension can be controlled by suture techniques: in non-layered suturing the xenograft acts as a super suture taking the tension away from the underlying native tissue by sliding it into place laterally, rather than pulling the fascial sheet sideways, resulting in tissue tension. |
The principles of regenerative vaginal surgery include: 1) transverse vaginal skin incisions and not vertical if possible; 2) dissections in tissue planes with stretching-counter stretching of tissues; 3) avoiding tension when suturing tissues; 4) using only non-inflammatory inducing monofilament suture materials; 5) refrain from using inflammatory inducing implants; 6) protect suture lines by bio-absorbable scaffolds, which also can support the ECM during remodeling.
If these principles are followed during primary surgery minimal scar tissue formation will result, with maximum return to normal regenerative native tissue. Minimal scar tissue formation provides an excellent surgical field should a secondary procedure is required: the patient will never be worse off from the primary surgeon’s surgical insult! In reconstructive terms the primary surgery will allow a foundation if further surgery may be needed - a staged surgical approach will be possible.
| Discussion | ▴Top |
“Diagnosis is not the end, but the beginning of practice”. Martin H. Fischer.
During the Kelly [19] and White [20] era, the only building blocks available to the surgeon were the tissues in front of him. Transfer of tissues from distant sites in the body - or from external sites - only became available late in the 20th century. These were called upon when the primary tissue was judged to be inadequate. This was replaced in due course by primary utilization of implant materials, ignoring the primary tissue as part of the “reconstructive” process. Surgeons with less experience in vaginal surgery were made to feel competent [21]. Complications with synthetic implants, FDA alert [22] and subsequent expensive lawsuits led to search for alternative techniques without use of any implant. Xenografts were also implicated in the debacle. A leap back to the previous comfort zone followed: the conventional colporrhaphy is back!
The time has come to reach beyond these “classical” native tissue procedures.
The secret of being a good physician is the ability to simplify a complicated problem. The simplistic manner, unfortunately, in which prolapse is being seen and treated by most, leads to the perfect storm we are experiencing at present [23]. Focus is being placed on reducing the symptom of a bulging vaginal wall, rather than looking for the origin of the defect that leads to the bulging wall. The art of healing surely involves a diagnostic workup looking for the origin of the disease that leads to the symptoms.
Tissue engineering is an interdisciplinary field that applies the principles of engineering and life sciences toward the development of biological substitutes that restore, maintain, or improve tissue function - or even replace whole organs [9, 24]. Regenerative medicine holds the promise of regenerating damaged tissues and organs in the body by replacing damaged tissue and/or by stimulating the bodies own repair mechanisms to heal previously irreparable tissues or organs [3]. With surgical repair of a wound or defect, continuity is established without regard to the exact replacement of lost or injured tissue. In contrast, regeneration is restoration with an exact copy with, not only morphologic continuity, but also functionality. This concept has been in clinical practise since 1997 [25]. Regenerative medicine is a recognized component of plastic surgery and other disciplines [12, 25, 26]. Principles of regenerative surgery, without using any synthetic material, had been applied to inguinal hernia repair with success [27, 28].
| Conclusion | ▴Top |
It is possible to apply the principles of regenerative surgery to vaginal wall prolapse surgery. This is especially important at this time with the controversies surround the use of synthetic mesh in prolapse repair. At this time one can return to the surgical techniques of the past, or improve them.
Native tissue is the primary building block in the reconstructive process. Knowledge of the template of normality, the anatomic supports of the vagina, which needs to be reproduced, is essential. Accessibility to this support system in its entirety is essential and is best achieved through the vaginal route. The defect in the vaginal support that leads to the prolapse can be detected and treated accordingly.
The first surgeon who attempts to repair the damage is the most important one. The surgical footprint left behind is not only paramount in the success of the primary surgery, but also may impede secondary surgical reconstructive attempts if the first procedure fails to give an optimal surgical outcome. The secondary procedure must not be an undo-redo process, but rather an add-on process. By following regenerative surgical principles minimal tissue damage occurs, and thus reduces difficulties with second surgery.
Regenerative surgery [12] puts the focus on the surgeon to do surgery on the “do-no-harm” principle. Rather than focusing on the size of the vaginal bulge, the cause of the bulge should be sought and repaired by following the principles of regenerative surgery. The surgeon’s ability to dissect, detect and repair the defect that led to prolapse will influence the surgical outcome. It is too unpredictable to rely solely on the ability of implant materials to give a successful surgical outcome. This can also be damaging to the patient. However, these materials may be utilized as an adjuvant to primary tissue healing in order to improve the outcome.
Grant Support
Nil.
Financial Disclosures
AN: none. CEA: travel bursary Kebomed UK. SM: travel bursary Cook Medical USA, Kebomed UK and Astellas.
| References | ▴Top |
- Davies J, editor, Tissue Regeneration - From Basic Biology to Clinical Application. InTech. 2012. Accessed 4 Jan 2015. Available from: http://www.intechopen.com/books/editor/tissue-regeneration-from-basic-biology-to-clinical-application.
- Kemp P. History of regenerative medicine: looking backwards to move forwards. Regen Med. 2006;1(5):653-669.
doi pubmed - Mason C, Dunnill P. A brief definition of regenerative medicine. Regen Med. 2008;3(1):1-5.
doi pubmed - Yannas IV, Kwan MD, Longaker MT. Early fetal healing as a model for adult organ regeneration. Tissue Eng. 2007;13(8):1789-1798.
doi pubmed - Diegelmann RF, Evans MC. Wound healing: an overview of acute, fibrotic and delayed healing. Front Biosci. 2004;9:283-289.
doi - Schultz GS, Ladwig G, Wysocki A. Extracellular matrix: review of its roles in acute and chronic wounds. 2005. Accessed January 2015. Available from: http://www.worldwidewounds.com/2005/august/Schultz/Extrace-Matric-Acute-Chronic-Wounds.html.
- Gattazzo F, Urciuolo A, Bonaldo P. Extracellular matrix: a dynamic microenvironment for stem cell niche. Biochim Biophys Acta. 2014;1840(8):2506-2519.
doi pubmed - van Neck J, Tuk B, Barritault D, Tong M. Heparan Sulfate Proteoglycan Mimetics Promote Tissue Regeneration: An Overview. In: Davies J, editor. Tissue Regeneration - From Basic Biology to Clinical Application. InTech, 2012. Accessed January 2015. Available from: http://www.intechopen.com/books/tissue-regeneration-from-basic-biology-to-clinical-application/heparan-sulfate-proteoglycan-mimetics-thrive-tissue-regeneration-an-overview.
- Langer R, Vacanti JP. Tissue engineering. Science. 1993;260(5110):920-926.
doi pubmed - Yannas IV. Tissue and Organ Regeneration in Adults. New York: Springer. 2001:1.
- Occleston NL, Metcalfe AD, Boanas A, Burgoyne NJ, Nield K, O'Kane S, Ferguson MW. Therapeutic improvement of scarring: mechanisms of scarless and scar-forming healing and approaches to the discovery of new treatments. Dermatol Res Pract. 2010;2010
- Orlando G, Wood KJ, De Coppi P, Baptista PM, Binder KW, Bitar KN, Breuer C, et al. Regenerative medicine as applied to general surgery. Ann Surg. 2012;255(5):867-880.
doi pubmed - Ferguson MW, O'Kane S. Scar-free healing: from embryonic mechanisms to adult therapeutic intervention. Philos Trans R Soc Lond B Biol Sci. 2004;359(1445):839-850.
doi pubmed - Ferguson MW. Wound healing--scar wars. Ulster Med J. 1998;67(Suppl 1):37-40.
pubmed - Villar CC, Cochran DL. Regeneration of periodontal tissues: guided tissue regeneration. Dent Clin North Am. 2010;54(1):73-92.
doi pubmed - Haggstrom M. "Phases of wound healing". "Medical gallery of Mikael Haggstrom 2014". Wikiversity Journal of Medicine 1 (2). Accessed January 2015. Available from: http://en.wikipedia.org/wiki/File:Wound_healing_phases.png#filelinks.
- Reid RI, Luo K. Site-specific prolapse surgery. II. Vaginal paravaginal repair augmented with either synthetic mesh or remodelling xenograft. Int Urogynecol J. 2011;22(5):601-609.
doi pubmed - Reid R. Hernia principles: what general surgeons can teach us about prolapse repair. In: Von Theobald P, Zimmerman CW, Davila W, eds. Vaginal prolapse surgery: new techniques. Guildford: Springer, 2011:19-39.
doi - Cooke TJ, Gousse AE. A historical perspective on cystocele repair--from honey to pessaries to anterior colporrhaphy: lessons from the past. J Urol. 2008;179(6):2126-2130.
doi pubmed - White GR. Cystocele--a radical cure by suturing lateral sulci of the vagina to the white line of pelvic fascia. 1909. Int Urogynecol J Pelvic Floor Dysfunct. 1997;8(5):288-292.
doi pubmed - Nieuwoudt A. Surgical footprints, then and now. Int Urogynecol J Pelvic Floor Dysfunct. 2008;19(9):1187-1188.
doi pubmed - FDA Safety Communication: UPDATE on serious complications associated with transvaginal placement of surgical mesh for pelvic organ prolapse. 2011. Available from: http://www.fda.gov/MedicalDevices/Safety/AlertsandNotices/ucm262435.htm.
- Brubaker L, Shull B. A perfect storm. Int Urogynecol J. 2012;23(1):3-4.
doi pubmed - Boennelycke M, Gras S, Lose G. Tissue engineering as a potential alternative or adjunct to surgical reconstruction in treating pelvic organ prolapse. Int Urogynecol J. 2013;24(5):741-747.
doi pubmed - Whitman DH, Berry RL, Green DM. Platelet gel: an autologous alternative to fibrin glue with applications in oral and maxillofacial surgery. J Oral Maxillofac Surg. 1997;55(11):1294-1299.
doi - International Society of Plastic Regenerative Surgery. Why regenerative surgery? 2014. Accessed December 2014. Available from: http://www.ispres-ipras.org/site/why-regenerative-surgery.
- Guarnieri A, Moscatelli F, Guarnieri F, Ravo B. A new technique for indirect inguinal hernia repair. Am J Surg. 1992;164(1):70-73.
doi - Desarda MP. Physiological repair of inguinal hernia: a new technique (study of 860 patients). Hernia. 2006;10(2):143-146.
doi pubmed
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Clinical Gynecology and Obstetrics is published by Elmer Press Inc.







