| Journal of Clinical Gynecology and Obstetrics, ISSN 1927-1271 print, 1927-128X online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Clin Gynecol Obstet and Elmer Press Inc |
| Journal website https://www.jcgo.org |
Case Report
Volume 10, Number 2, June 2021, pages 55-58
A Case of a Rare Malignant Mixed Ovarian Germ Cell Tumor Masquerading as an Ectopic Pregnancy
Ria Marwahaa, Panagiotis Giamougiannisb, Raj Naikc, Helen F. Stringfellowd, Antonios Anagnostopoulosb, Gemma L. Owensa, b, e
aFaculty of Medicine, Biology and Health, The University of Manchester, Oxford Road, Manchester, M13 9PL, UK
bGynaecological Oncology, Lancashire Teaching Hospitals NHS Trust, Royal Preston Hospital, Sharoe Green Lane, Fulwood, Preston, PR2 9HT, UK
cGynaecological Oncology, Queen Elizabeth Hospital, Gateshead, NE9 6SX, UK
dPathology, Lancashire Teaching Hospitals NHS Trust, Royal Preston Hospital, Sharoe Green Lane, Fulwood, Preston, PR2 9HT, UK
eCorresponding Author: Gemma L. Owens, Gynaecological Oncology, Division of Cancer Sciences, School of Medicine, Faculty of Medicine, Biology and Health, The University of Manchester, Research Floor, St Mary’s Hospital, Oxford Road, Manchester, M13 9WL, UK
Manuscript submitted April 15, 2021, accepted June 14, 2021, published online June 24, 2021
Short title: Malignant Mixed Ovarian Germ Cell Tumor
doi: https://doi.org/10.14740/jcgo743
| Abstract | ▴Top |
Malignant ovarian germ cell tumors (MOGCTs) are rare but disproportionately affect young women and adolescent girls. Although clinical presentation varies, these tumors can present with an adnexal mass and raised serum beta-human chorionic gonadotrophin (β-hCG), mimicking an ectopic pregnancy. We present the case of a 20-year-old nulliparous woman, who was initially treated both medically and surgically as an ectopic pregnancy, and was eventually diagnosed with a malignant mixed germ cell tumor (MMGCT) following laparoscopy. We highlight the diagnostic challenges of ectopic pregnancies versus MOGCTs, and the importance of adopting a fertility-sparing approach in young nulliparous women.
Keywords: Germ cell tumors; Ovary; Surgery; Chemotherapy; Gynecological cancers
| Introduction | ▴Top |
Malignant ovarian germ cell tumors (MOGCTs) are rare, accounting for less than 5% of all ovarian cancers [1]. This heterogeneous group of tumors derives from primitive ovarian germ cells and includes dysgerminomas, immature teratomas, yolk sac tumors, embryonal carcinomas, choriocarcinomas and mixed MOGCTs [2]. Although they can present at any age, they disproportionately affect young women and adolescent girls. Typically, MOGCTs present with abdominal pain in the presence of an adnexal mass [2]. In addition, some germ cell tumors, specifically choriocarcinomas, embryonal tumors, mixed germ cell tumors and dysgerminomas, secrete beta-human chorionic gonadotrophin (β-hCG) [1]. This triad of findings can also be indicative of an ectopic pregnancy, making the diagnosis of this rare tumor challenging in women of reproductive age. We present a case of a young nulliparous woman with a malignant mixed germ cell tumor (MMGCT), initially misdiagnosed as an ectopic pregnancy.
| Case Report | ▴Top |
A 20-year-old nulliparous woman initially presented to our subfertility clinic with a history of polycystic ovarian syndrome. She had experienced menarche at the age of 12 and reported having heavy and irregular periods. She was not using any contraception as she was trying to conceive, and her last menstrual period was approximately 7 weeks earlier. She was found to have a positive pregnancy test and serial β-hCG blood tests were arranged via the Early Pregnancy Assessment Unit. She reported no local or systemic symptoms at initial presentation. Serial β-hCG levels plateaued at 229 IU/L and a transvaginal ultrasound scan showed a 3.1 cm left adnexal mass without free fluid in the pelvis.
The patient was counseled regarding the risks of an ectopic pregnancy, and offered the options of either medical management with methotrexate or surgical management with laparoscopy. The patient opted for medical management and 80 mg methotrexate was administered at a baseline β-hCG of 251 IU/L. Initially, β-hCG levels showed an optimal decrease with more than 15% drop between day 4 and 7 following methotrexate; however, the level increased to 287 IU/L on day 14. According to protocol, the patient was invited back for further assessment but on review was asymptomatic. A repeat ultrasound scan showed a left hematosalpinx and an enlarging left adnexal mass, now measuring 4.3 cm. In view of an increasing adnexal mass, the patient was taken to theatre for surgical management of a suspected ectopic pregnancy. Laparoscopic findings (Fig. 1) revealed a left hydrosalpinx and a vascular ovarian cystic mass on the surface of the left ovary, presumed to be an ovarian ectopic pregnancy, which was densely adherent to the left paracolic peritoneum and fat. The ovary was mobilized from the pelvic side wall and the presumed ectopic pregnancy was easily dissected leaving the majority of the left ovary intact. The left hydrosalpinx was also excised, while the rest of the pelvis and abdomen were noted to be normal.
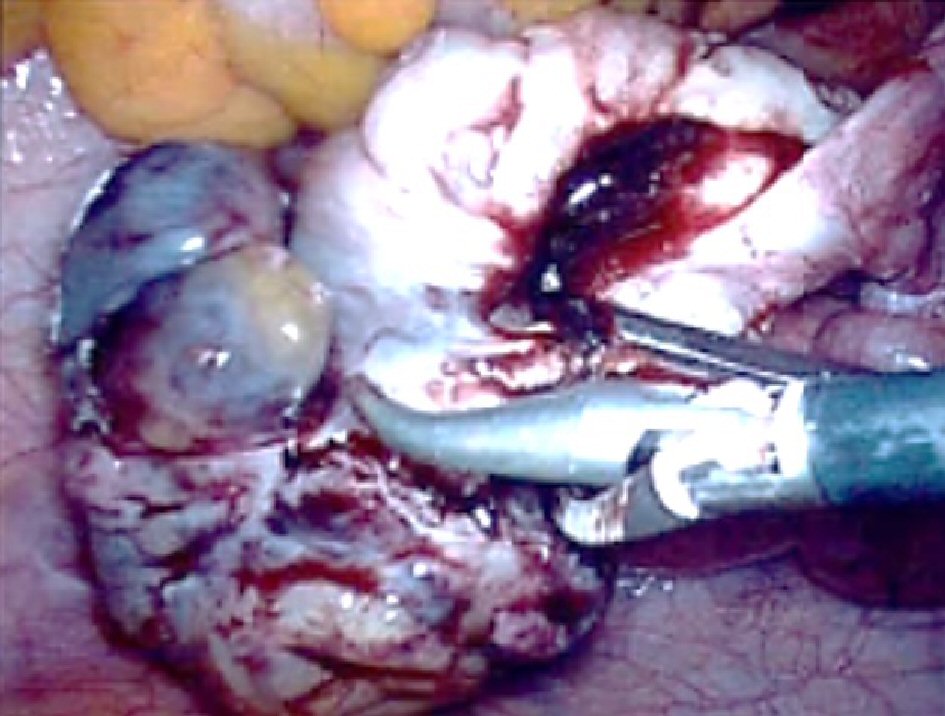 Click for large image | Figure 1. Laparoscopic view of the left ovarian mass presumed to be an ovarian ectopic. |
Final histology demonstrated a MMGCT with 40% embryonal cell carcinoma, 40% yolk sac tumor, 15% teratoma and 5% choriocarcinoma components (Fig. 2).A staging computed tomography (CT) scan of the thorax, abdomen and pelvis showed no evidence of disseminated disease and an International Federation of Gynecology and Obstetrics (FIGO) stage IC tumor was assumed. Following discussion at the tumor board meeting, the patient was referred for adjuvant chemotherapy in the form of bleomycin, etoposide and cisplatin (BEP). She received three 3-weekly cycles of etoposide 165 mg/m2 given on day 1, 2 and 3, cisplatin 50 mg/m2 given on day 1 and 2, and bleomycin 30 IU given on day 2, 8 and 15 of each cycle.
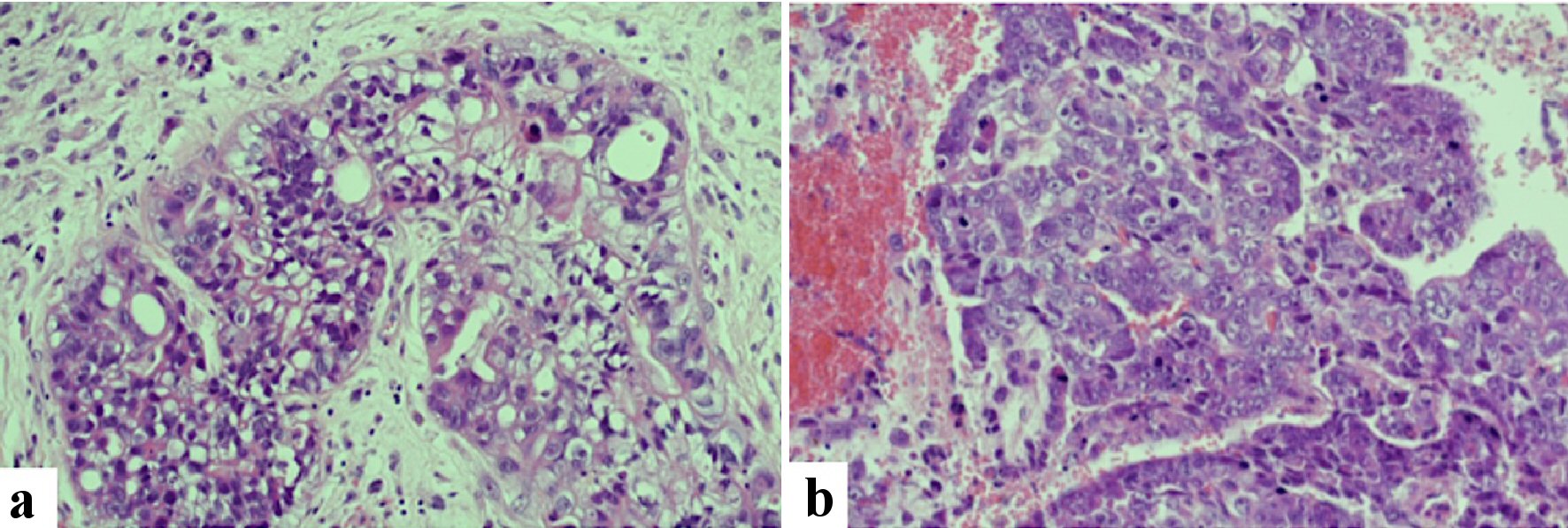 Click for large image | Figure 2. Histopathological assessment identified a malignant mixed ovarian germ cell tumor. Yolk sac tumor (a) and choriocarcinoma (b) components by H&E staining are shown. Original magnifications are × 100. H&E: hematoxylin and eosin stain. |
The patient was offered and underwent fertility preservation with oocyte retrieval prior to commencing chemotherapy. A few days later she required hospital admission due to severe ovarian hyperstimulation syndrome with the presence of ascites and pleural effusions on CT scan. The initiation of chemotherapy was not delayed; however, she completed the first cycle as an inpatient due to severe neutropenia. There was also deterioration in her liver function, which led to omission of one dose of etoposide. Her clinical picture gradually improved and she was discharged from the hospital at the end of her first cycle. The rest of her chemotherapy course was completed without disruptions and without the need for further hospitalization.
Upon chemotherapy completion, a further CT scan of her chest, abdomen and pelvis demonstrated a slight increase in the volume of her right ovary, but the pleural effusions and ascites had resolved. Following advice by the regional tumor board, she underwent a laparoscopic left oophorectomy and omental biopsy 2 months after the last cycle of chemotherapy, in order to complete staging of her malignancy. No residual disease was noted intraoperatively and on histological analysis of excised tissues. She had another CT scan 3 months following her second operation, which showed stable size of her right ovary and no recurrent or metastatic disease. She did not receive any further chemotherapy or radiotherapy and is currently under 3-monthly follow-up. One year following initial diagnosis, the patient remains well with no signs of recurrence.
| Discussion | ▴Top |
Gynecologists often overlook a differential diagnosis of an ovarian germ cell tumor in young women presenting with elevated β-hCG and an adnexal mass, given the relative frequency of ectopic pregnancy [3]. On reviewing the literature, we identified seven similar reports of MOGCTs mimicking an ectopic pregnancy (Table 1) [3-9]. In all cases, the true diagnosis was only revealed on histological examination of the tissue. Six cases were initially managed surgically, via laparoscopy or laparotomy [3-8]. One case was initially managed with methotrexate, only to revert to laparoscopy after failure to achieve drop in β-hCG levels after repeated doses [9]. To the best of our knowledge, this is the first case of a MMGCT diagnosed after failed initial medical management of a presumed ectopic pregnancy. MMGCTs consist of at least two germ cell components and represent less than 1% of all ovarian germ cell tumors; those containing yolk sac tumors, high-grade immature teratomas and embryonal carcinomas, are associated with a more aggressive phenotype [10].
 Click to view | Table 1. Comparison of Clinical Findings and Management of Malignant Ovarian Germ Cell Tumors Presenting as Apparent Ectopic Pregnancies |
Most MOGCTs are unilateral and as such, fertility-sparing surgery with unilateral oophorectomy and surgical staging is recommended in women with apparent early disease [2]. In the case reported here, a conservative approach at the initial surgery was adopted, given the patient’s young age, future fertility desires and the presumed diagnosis of an ectopic pregnancy. The partial oophorectomy performed not only led to surgical spill but most likely allowed microscopic residual tumor to remain onto the ovarian surface. Despite this management, following chemotherapy neither second-look laparoscopy nor radiology detected macroscopic residual tumor, while histopathology and biochemical markers were not indicative of residual microscopic disease. The tumor involved the surface of the left ovary hence rendering it FIGO stage IC2 prior to surgical spill. Although there is now a growing body of evidence for a “surveillance only” strategy after surgical resection of FIGO stage IA/B MOGCTs [11, 12], adjuvant chemotherapy is recommended for FIGO stage IC tumors. As such, the treatment plan was not altered by the surgical spill and partial oophorectomy, and ultimately, it is unlikely to have significantly affected the patient’s long-term prognosis.
Laparotomy is currently advocated for cases of MOGCTs, owing to the solid nature and large size of these tumors [13]. Data regarding laparoscopic management of MOGCTs are scarce. A single centre study of 28 patients, who underwent laparoscopic surgery for malignant non-epithelial ovarian tumors which included MOGCTs, concluded that this approach was feasible and safe without compromising survival [13]. We offered our patient laparoscopic surgery to complete her treatment, as the residual ovarian tissue remained small size on imaging; there was no overt invasive disease and the risks of laparoscopy, including possible peritoneal tumor dissemination and port-site metastasis, were lower compared to the initial operation.
MOGCTs predominantly affect young women, many of whom will be nulliparous and therefore fertility preservation should be a priority in the management of these tumors. Adjuvant BEP chemotherapy following fertility-preserving surgery has been associated with good long-term survival outcomes and no apparent ovarian dysfunction or failure in non-dysgerminomatous MOGCTs [12, 14, 15]. As a precaution, given that our patient first presented via the subfertility service, she underwent oocyte retrieval prior to commencing chemotherapy. In such instances, early involvement of fertility experts is required to enable oocyte cryopreservation, in a timely manner, so as not to delay treatment of the cancer.
Although diagnosis of an ectopic pregnancy is by far the commonest amongst women of reproductive age presenting with an elevated serum β-hCG level and a pelvic mass, the possibility of a MOGCT should be considered, particularly if a patient fails to respond to medical management. As the majority of women affected by MOGCTs are young and desire fertility preservation, it is important to adopt a conservative surgical approach and consider early involvement of fertility experts. Although data supporting laparoscopic management of MOGCTs are limited, our case highlights that minimal access surgery can be considered in carefully selected cases.
Acknowledgments
We would like to thank the Histopathology Department for their assistance in selecting histopathology section images for inclusion in this report.
Financial Disclosure
None to declare.
Conflict of Interest
The authors have no conflict of interest to disclose.
Informed Consent
Written informed consent was obtained from the patient for publication of this case report and accompanying images.
Author Contributions
RM, PG and GLO were responsible for drafting and editing the report. AA, HFS, RN and GLO were directly involved in the care of the patient described in this case report.
Data Availability
The authors declare that data supporting the findings of this case report are available within the article.
| References | ▴Top |
- Parkinson CA, Hatcher HM, Earl HM, Ajithkumar TV. Multidisciplinary management of malignant ovarian germ cell tumours. Gynecol Oncol. 2011;121(3):625-636.
doi pubmed - Low JJ, Ilancheran A, Ng JS. Malignant ovarian germ-cell tumours. Best Pract Res Clin Obstet Gynaecol. 2012;26(3):347-355.
doi pubmed - Rozenholc A, Abdulcadir J, Pelte MF, Petignat P. A pelvic mass on ultrasonography and high human chorionic gonadotropin level: not always an ectopic pregnancy. BMJ Case Rep. 2012;2012:bcr0120125577.
doi pubmed - Kucera C, Cox-Bauer C, Miller C. Apparent ectopic pregnancy with unexpected finding of a germ cell tumor: A case report. Gynecol Oncol Rep. 2017;21:31-33.
doi pubmed - Ozkaya O, Sezik M, Aydin AR, Kapucuoglu N, Kaya H. Mixed germ cell tumor of the ovary mimicking unruptured ectopic pregnancy presenting with unusually high serum alpha-fetoprotein level. Gynecol Surg. 2005;2:307-310.
- Dawley B, Acuna A, Grasu B. Ectopic production of HCG by a benign ovarian mature cystic teratoma simulating an extra-uterine pregnancy: a case report. W V Med J. 2012;108(1):15-17.
- Balat O, Kutlar I, Ozkur A, Bakir K, Aksoy F, Ugur MG. Primary pure ovarian choriocarcinoma mimicking ectopic pregnancy: a report of fulminant progression. Tumori. 2004;90(1):136-138.
doi pubmed - Freij MA, Saleh H, Hamouda T, Raje G, Saleh M. Primary ovarian choriocarcinoma presenting with acute abdomen mimicking an ectopic pregnancy. Eur J Gynaecol Oncol. 2011;32(4):425-426.
- Horne AW, Sutherland S, Faratian D, Farquharson DI, Duncan WC. Ovarian choriocarcinoma masquerading as ectopic pregnancy. J Obstet Gynaecol. 2006;26(4):385-387.
doi pubmed - Lai CH, Chang TC, Hsueh S, Wu TI, Chao A, Chou HH, Wang PN. Outcome and prognostic factors in ovarian germ cell malignancies. Gynecol Oncol. 2005;96(3):784-791.
doi pubmed - Patterson DM, Murugaesu N, Holden L, Seckl MJ, Rustin GJ. A review of the close surveillance policy for stage I female germ cell tumors of the ovary and other sites. Int J Gynecol Cancer. 2008;18(1):43-50.
doi pubmed - Weinberg LE, Lurain JR, Singh DK, Schink JC. Survival and reproductive outcomes in women treated for malignant ovarian germ cell tumors. Gynecol Oncol. 2011;121(2):285-289.
doi pubmed - Shim SH, Kim DY, Lee SW, Park JY, Kim JH, Kim YM, Kim YT, et al. Laparoscopic management of early-stage malignant nonepithelial ovarian tumors: surgical and survival outcomes. Int J Gynecol Cancer. 2013;23(2):249-255.
doi pubmed - Low JJ, Perrin LC, Crandon AJ, Hacker NF. Conservative surgery to preserve ovarian function in patients with malignant ovarian germ cell tumors. A review of 74 cases. Cancer. 2000;89(2):391-398.
doi - Tangir J, Zelterman D, Ma W, Schwartz PE. Reproductive function after conservative surgery and chemotherapy for malignant germ cell tumors of the ovary. Obstet Gynecol. 2003;101(2):251-257.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Clinical Gynecology and Obstetrics is published by Elmer Press Inc.

